
Look around. That device in front of you. The seat beneath. Even the invisible stuff filling your lungs. All of it pieces together from specks too small to see - atoms, they’re called. Now picture one of those bits. What lives inside? Could there be something smaller still?
Atoms once seemed like nature's tiniest building blocks. That idea hides in the word itself - rooted in ancient Greek for "uncut-able." Today’s findings tell a different story though. These tiny units aren’t hard spheres after all. Smaller pieces live inside them, revealed by careful study.
A tiny world waits when you step past the surface of matter. Inside each bit, movement never stops. One speck holds vast space between pieces that buzz like fireflies. Hidden corners glow with energy others miss. What seems solid turns restless up close.
A tiny center sits inside every atom. This core connects to a cloud of particles that zip around it
A single speck, much like a miniature version of space's grand design. Inside, at its heart, something dense takes the central spot - much as sunlight dominates our skies. Whirling outward, smaller pieces trace paths without touching, held close yet always distant.
Inside each atom sits a tiny core. Though the whole atom is minuscule, its heart takes up far less space - yet nearly all the weight lives there.
Deep within the core of an atom sit a pair of key components
Atoms get their identity from how many protons they carry. Take hydrogen - just one proton makes it what it is
Inside the atom's core, neutrons act like glue. When missing, proton forces push everything apart - structure fails fast. Stability depends on these neutral particles staying put.
Speeding through space beyond the core, electrons exist as small bits with negative charge. Around the central hub they race, these specks never still.
Here's a twist: electrons avoid tidy orbits entirely. Instead of circling like tiny planets, they hang around in fuzzy zones scientists call clouds. Within these areas, it is impossible to pin down their exact spot - only guess where they might show up. Quantum rules shape this behavior, making location a game of odds.
Electrons are responsible for:
It's the tiny particles that make reactions possible - no electrons, no chemical activity at all.
Picture this. Atoms contain far more nothing than stuff. Space fills most of what you see. What feels solid is nearly hollow. Almost everything around you? Built on gaps.
Picture a cricket ball sitting at the center of a huge stadium. Around its farthest seats, tiny specks zip unpredictably along the rims. Those specks are electrons, circling distant boundaries. The vast stretch between center and edge holds nearly nothing at all. Empty gaps dominate what we call solid matter.
Still, it’s the push and pull among tiny particles that makes objects seem firm to our touch.
Turns out, what we thought were basic building blocks - protons and neutrons - are actually built from something tinier. Quarks make up those particles, hiding one level deeper inside.
Inside the nucleus, quarks stick close thanks to intense pushes and pulls. Huge devices, such as particle accelerators, let scientists peek at these tiny bits.
At its core, this area dives into quantum physics - offering a window into how tiny pieces shape everything. Sometimes it feels like puzzle work, yet each discovery quietly reveals more about nature's foundation.
So, what is inside an atom?
Right in the middle sits a cluster of protons and neutrons called the nucleus. Zoom out slightly, electrons zip around it, too quick to pin down, creating what looks like a fuzzy shell. Beneath that surface, within each proton and neutron, tiny pieces named quarks take up space.
Inside something so small you cannot spot it lies answers about what stuff is made of. Though invisible, each one carries clues about how things join, break apart, react. A speck like this tells stories of heat, light, change, space far beyond us. Hidden within its core rests knowledge stretching into stars and time.
Hold an object sometime soon. Inside, countless atoms hum along - each one hides a small, strange universe within. Their inner lives pulse quietly, full of odd detail.
Ever seen how a tree, a bird, wind, rain, and dirt somehow fit together? That web - how life links to surroundings - is what folks mean by ecology. It shows creatures shaping one another’s lives while sharing air, ground, or rivers.
Biology holds a piece of the puzzle - this part shows why nature doesn’t tip too far one way.
Home. That is what the Greek term oikos stands for, where "ecology" finds its roots. Living things do not exist alone; they tie into environments around them. This connection forms the core of ecological science. A planet breathes, shifts, holds life - it functions like any dwelling, shaped by those inside and outside its walls.
It includes the study of:
Ecology focuses on questions like:
A single tree might feed a squirrel while giving it a place to rest. Yet when that animal moves, seeds travel far beyond their starting point. Once life ends, tiny organisms take apart what remains. From decay, fresh nourishment flows into the ground below. Each piece connects - this is how ecology works.
Ecology can be studied at different levels:
Ecology helps us:
Lives here could falter if nature's rhythm stumbles. Balance keeps things ticking without loud fanfare.

Living things don’t exist alone - ecology looks at how they link with one another, along with the world around them. This web of life reveals balance, where change in one spot can ripple through everything else.
What happens when we grasp how nature works? We start seeing why safeguarding the planet matters. Sure, there's just one Earth - a fact that makes care for it less optional. Balance doesn’t happen by accident; people have to maintain it.
One light might obey just a single switch. Another could respond to taps from spots far apart. That behavior comes down to what kind of switch lives inside the wall. You will usually find either a 1 way or a 2 way version doing the job.
Grasping how these switches work matters when learning about everyday circuits and household connections. Here's a clear look at what sets them apart.
One spot only - that is where a basic switch does its job. From just one place, lights turn on or off. This kind works everywhere people live.
A single switch in a tiny space often controls the light just once. Hit it to spark the bulb, then tap it later to kill power - this setup lets you manage things from only one spot. Sometimes called a basic toggle, it responds each time without extra paths or remote triggers.
One terminal connects when the switch is off, another takes over once it flips on. Each position holds its own contact point without sharing. Power moves through one path at a time, never both together
Filled with current, the circuit wakes up once the switch clicks into place. Power moves through it now, alive.
A single lamp can be turned on or off from two spots using a setup with dual controls. Location does not matter when flipping the state of the bulb thanks to linked switching units.
A single light, like one on a stairway, might get powered up from downstairs then shut down upstairs. That kind of control works thanks to what's called a two-way switching setup.
From opposite ends of a room, control begins. Two switches link in tandem, forming a shared route. This setup shifts electricity’s direction through wiring. Light turns on or off at one location, then the other decides again. Movement across spaces meets response without extra parts. The flow reroutes each time a toggle moves. Control lives in both places, never just one.
Few changes show up right away once you look inside. Wiring sets them apart, yet what they do matters just as much.

A single-pole switch plays a key role in everyday wiring setups. Though basic in design, it handles straightforward on-off tasks reliably. Control from separate spots becomes possible thanks to its two-location operation. The second type, known for dual-point access, adds ease without complexity.
It becomes clearer how homes get wired once we see the distinction. Understanding this sets the foundation for grasping simple electricity ideas.
Picture an animal trying to survive out here. Sunlight feeds green plants, which are then eaten by creatures like rabbits. Those small grazers might become prey for hunters such as foxes. Each part ties into the next, forming what scientists name a food chain.
One creature eats another, passing energy along. See how life connects through eating? A plant gets eaten by a bug, that bug feeds a bird. Watch nature share power, bite by bite. Each meal shifts energy up, from small to bigger. Feel the flow when one being fuels another. This path of feeding links everyone alive.
Plants kick off the whole process by turning sunlight into food. From there, creatures start feeding on one another, passing energy along. One animal eats a plant, then gets eaten itself - this moves life fuel forward. What you see is just steps in survival, each linked through meals.
Plants grab energy from sunlight, kicking off each food chain. Without sunshine, they cannot grow. That growth feeds others down the line. Sunlight kicks everything into motion.
Green plants create food all by themselves. Sunlight fuels this change inside leaves, known as photosynthesis.
Examples:
Green life kicks off each meal path in nature.
Hungry creatures must eat because they can’t grow meals like plants do. Instead, they rely on green life or hunt fellow animals.
Folks who buy things fall into three groups
Fungi and bacteria chew through lifeless trees plus creatures, turning them into basic materials. Back into earth go these nourishing bits, ready again.
Examples:
A world without breakers-down turns into a graveyard of fallen trees and lifeless creatures. Dead things pile up when nothing tears them apart. Imagine forests choked by old bones and dried leaves. Rotting bodies stack higher each day if no small workers chew through the mess. Life depends on tiny eaters cleaning what others leave behind.
Sun feeds grass that deer eat which lions consume
In this food chain:
From plant to creature, energy travels forward. One life passes power to the next without pause. Step follows step in quiet transfer. Living forms link through this flow. Each eater takes what was stored before. Movement happens slowly, yet never stops. What fuels one becomes fuel elsewhere.
When a piece goes missing from a food chain, everything else wobbles. Nature stays steady because each eater depends on another below them.

From sunlight, energy moves through green life first. Plants pass it along when creatures eat them instead. Each eater fits into a bigger picture somehow. Living things that break down waste help recycle what others leave behind. Every part connects, even if quietly.
Every creature ties into a web much bigger than itself. Seeing who eats whom shows children just how deeply life links together out there.
Right now, dirty environments are a major problem across Earth. With factories spreading and urban areas getting bigger, toxins fill up the sky, rivers, and soil faster every year. Breathing bad air or touching contaminated ground hurts people, creatures, living forests - whole systems feel the impact. What we leave behind settles into life itself.
Start by seeing pollution as harm to nature around us. One kind shows up in dirty air, another fills water with trash. Think of soil soaked in chemicals, noise that never quits. Each part connects - smoke leads to rain trouble, plastic piles where birds nest. Ways out? Less waste matters. Choices slow damage when people act. Solutions grow where habits shift quietly.
Pollution shows up when unwelcome stuff gets into nature and throws things off track. That troublesome material? It goes by the name of pollutants. Sometimes it's a sludge, other times a vapor - or even something you could hold in your hand.
Fumes taint air, water, soil - life struggles where toxins spread. Nature's balance breaks when waste slips into rivers, fields, skies.
Fumes rise when cars run, factories operate, coal burns, or wildfires spread across woodlands. Gases such as CO₂ and SO₂ taint what we breathe every day.
Breathing trouble might show up, sometimes along with asthma or different health concerns. Sometimes it triggers asthma attacks, followed by other physical reactions too.
Trash from factories, human waste, along with toxic substances often flow into streams, ponds, or seas. Creatures living underwater suffer, while people can’t rely on that water to drink.
Farmers struggle when trash lingers too long underground. Plastics, spilled chemicals, or dumped refuse mess up earth meant for growing food. This damage steals nutrients plants need. Crops either shrink in number or fail completely.
From honking cars to factory machines, city life brings constant sound. Because of this, people often feel tense without knowing why. Machines blaring during the day might leave ears ringing later. When nights are noisy, rest becomes hard to find.
Major causes include:
We can reduce pollution by:

Fumes fill skies, rivers carry waste - life pays the price. Where land turns toxic, creatures struggle to survive.
Every little choice adds up when it comes to caring for nature. What we do today shapes how things grow tomorrow.
These days, trash handling in India stands out as a key environmental challenge. As more people move to cities and buy more things, garbage piles grow faster than ever before. Because of this pressure, dealing with refuse wisely helps keep communities safe and nature intact. What happens on streets and dumps affects air, water, even food over time.
Waste handling across India? It looks different depending on where you are. Some cities sort trash, others dump it without rules. Old habits slow progress, yet new methods pop up now and then. Money moves unevenly - some areas get gear, some don’t. People try fixing things, but systems often lag behind. Progress crawls, though small wins appear quietly.
India generates different types of waste, including:
From all of it, trash collected in neighborhoods and shopping areas makes up most. Though often overlooked, household scraps mixed with market leftovers pile up fast. What begins small grows heavy when gathered block by block. Daily life spills into bins more than expected. Streets fill where collection lags behind routine.
Waste Management In India
From homes, trash splits into rotting kinds and stuff that won’t break down. When sorted right where it’s made, turning things back into new materials gets simpler.
From old bottles to used cans, stuff once tossed away now gets another life through sorting and reuse. Plastic, paper, metal - each type follows its own path after pickup. Glass jars might become new containers instead of piling up in dumps. What was trash can turn into something useful again. Less ends up buried when these items get processed properly.
From kitchen leftovers to garden gold - rotten bits of fruit, wilted greens, even coffee grounds transform slowly. Not trash anymore, these materials break down into dark, earthy matter instead. Farmers toss it into soil; plants grow stronger because of it. A peel today feeds a tomato plant tomorrow.
Out in the open, non-recyclable trash often ends up buried underground. Still, piling too much waste into these sites harms nature around them.
Fuel made from trash comes from certain greenery, cutting down how much garbage piles up. Waste gets turned by some leafy things into power instead of rotting in dumps.
India faces several waste management challenges:
Fumes rise when trash burns without care, tainting skies above. Rain carries filth into streams, poisoning flow beneath. Earth cracks under waste that won’t break down, spreading harm below.
Folks started picking up brooms more often once Swachh Bharat kicked in. From streets to lanes, trash now finds its way into bins instead of drains. Some towns go further - turning food scraps into soil helpers behind parks and schools. Recycling centers pop up where dumps used to stink.

Fixing trash problems across India sounds tough - yet entirely possible. Sorting waste right, reusing materials, turning food scraps into soil, along with teaching people clearly, helps cut down dirt and build fresher surroundings.
A single person might seem small, yet their choices add up. Tossing trash into separate bins where you live - this act shapes what kind of country we get.
Underneath your feet lies something powerful - India's soil, a quiet backbone of life here. Not just farms but entire forests depend on it, along with countless people who earn their daily bread. Thanks to its rich mix of types, the land grows everything: grain after grain, leafy tea bushes, fluffy cotton fields. What you see growing across regions comes from what runs deep below.
Let’s understand the major types of Indian soil and why they are important.
From snowy peaks down to tropical coasts, India's land shifts underfoot. Where rain pours heavy, earth turns red and rich. In dry western stretches, sand mixes with pale, lifeless dirt. River valleys pile up soft silt after each flood passes through. Highlands wear dark cotton soils cracked wide in heat. Coastal strips hold salty traces left by tides long gone. Each region shapes what lies beneath it slowly
River-made dirt covers big parts of India. This kind spreads wide across the north where water flows through flat lands. Ganga and Brahmaputra shaped these areas over time. Rich earth grows plenty here because of how it was laid down.
Filled with nourishing elements, this soil supports strong growth of rice, alongside wheat that thrives just as well. Sugarcane follows, doing well where pulses also take root easily.
Dark earth goes by another name - regur soil. Across the Deccan Plateau, that’s where it settles most.
Moisture stays put here, making it work well when planting cotton. Soybean thrives too, since the earth holds what rain leaves behind. Groundnut fits just right, thanks to how damp things stay below.
Down south and east across India, you see red soil stretching out. Iron oxide gives it that reddish look, nothing else does.
Crops such as millets grow well under these conditions, while groundnuts also thrive. Pulses fit into this pattern too, doing best when similar needs are met.
Fine red earth takes shape where rains pour hard under hot sun. On rising lands, this ground shows up most often.
Fed well, this soil holds up tea plants, alongside coffee, even cashew trees.
Sometimes sand holds almost no water, seen across places such as Rajasthan. Lacking rich plant leftovers, it still grows food when given steady watering.
Indian soil plays a major role in:
Farm types shift with soil kinds across India's land. That variety feeds a wide mix of growing needs.

Fertile alluvial stretches across flat lands, feeding much of what grows. Yet on high ground, dark soils hold water well, supporting different crops through dry spells. What rises from earth shapes more than harvests - it guides livelihoods. Each patch below our feet plays a part, though often unseen.
Soil in India tells a story of life, quietly feeding countless families through what it gives to crops year after year. What grows depends on what lies beneath, shaping choices far beyond farm borders.
What if animal-made wonders could rival human construction? The biggest beaver dam on Earth gives a hint. Not a single person helped build it. Instead, busy rodents shaped this giant creation. Nature often surprises when left to its own rhythm. This quiet powerhouse stands far from cities. A slow process turned trees into an engineered landscape. Life builds in ways we rarely expect.
Small though they are, beavers craft with surprising skill. Take a look at where this particular dam sits - its place tells a story. What makes it stand out isn’t just size, but how it shapes the land around it.
Hidden deep in the wilderness of northern Canada sits a giant beaver creation. Found by chance on satellite photos back in 2007, it stretches far beyond what anyone expected. Located within Wood Buffalo National Park, this enormous dam reshapes how we see animal-built structures.
Picture eight full-sized pitches lined up one after another - that gives you an idea of the dam’s reach. Running close to 850 meters, it covers almost half a mile across the landscape.
Beavers are often called “nature’s engineers.” They build dams using:
Water slows when beavers block it with barriers made of sticks. Ponds form behind these obstructions - places where they shape dome-like shelters. Protection comes from the wet area around those dens, keeping threats such as bears at a distance.
Built slowly, the biggest beaver dam took shape across decades. Different families added parts, each generation extending what came before.
Beaver dams are extremely important for the environment. They:
Built by creatures, the structure holds up a full web of life, boosting variety in nature around it.

Built by busy paws under open skies, the longest beaver dam on Earth stretches across wilderness land. In northern Canada it rests - a half-mile ribbon of sticks shaped by quiet persistence instead of plans.
Bold architects? Try watching a beaver at work. Next moment you're picturing big steel frames, recall - nature's heavy lifters shape landscapes too. Not with cranes, mind you, but teeth and mud and stubborn patience. When humans dream up grand designs, these rodents have already dammed the river. Their blueprint comes from instinct, not blueprints. Impressive doesn’t begin to cover it.
A hands-on task in class often sparks curiosity like nothing else. Through it, young learners step outside pages filled with facts. Doing rather than only studying lets them test concepts by mixing chemicals or shaping prototypes. Watching reactions unfold shows exactly where theory meets reality.
Curiosity grows when hands-on work takes center stage. A project built step by step teaches more than theory alone ever could. Because thinking differently matters, trying out ideas becomes natural. When questions lead to testing, learning sticks deeper. Success often comes from small fixes made along the way. Starting simple keeps things clear. One idea at a time shapes strong results.
Science projects encourage students to:
Finding answers through hands-on tasks helps pupils grasp science better. Doing things themselves sticks in their minds more than just listening.
Try these thoughts on for size. A few notions that might stick around awhile. Ponder a moment longer than expected. Watch how they unfold without rushing. Notice what comes up when you least expect it
Starting off simply, these tasks show science ideas in a clear way. With little setup needed, each one makes learning visible through doing.

A science fair task isn’t only about turning in work. Curiosity opens doors to testing ideas through hands-on discovery. When imagination guides preparation, results often surprise even the maker.
A spark shows up once hands dive into doing real things. That moment? A solid science activity makes it happen.
Most people never think twice about turning on a tap. Yet that clear glass of water started out somewhere murky. Imagine rainwater rushing through soil, picking up mess along the way. From ponds and streams, it travels toward hidden systems underground. A facility steps in before any bottle gets filled. Pipes carry wild liquid into quiet chambers below ground. Chemicals nudge dirt loose, making particles stick together. Gravity pulls muck downward while cleaner fluid rises. Filters made of sand grab what remains unseen. Finally, tiny invaders meet their match - zapped or blocked completely. Only then does flow earn its place at your sink.
Water gets cleaned at a treatment facility by taking out particles, dangerous substances, one thing after another - tiny organisms included - to make it fit for people to use. Step follows step in sequence; here is how each part unfolds.
From rivers, lakes, or reservoirs, water gets pulled into the system. Still, it carries along mud, sand, bacteria, yet plenty of unseen junk. Because of that, cleaning steps happen before it ever arrives at taps.
During this phase, substances such as alum go into the water. Because of them, tiny bits of dirt begin grouping into bigger masses known as flocs.
After that, stillness takes over inside big holding tanks. Down below, the dense clumps begin to drop. That dropping phase has a name - sedimentation. Heavy bits stay put once they hit the floor.
Floating on top, the clean water moves forward into what comes next.
Through layers of sand, gravel, then charcoal, water moves slowly. Tiny bits left after sedimentation get caught in these filtering stages.
Filtration takes out nearly all particles you can see.
Filtration does not always remove every dangerous germ. Because of this, substances such as chlorine go into the water afterward - stopping microbes by breaking them down.
Only after this happens can people drink the water without risk. Water becomes fit to consume once this process finishes. Safety comes when this stage completes properly. This part makes sure no harm hides in each sip later. When done right, every glass stays clean and harmless.
After that comes storage - clean tanks hold the water until it flows out through pipes, reaching houses, classrooms, schools, factories.

Water gets cleaned by moving through a series of stages: first it's gathered, then chemicals are added so tiny particles stick together. After that, those clumps settle out on their own. Next, the liquid slips through layers of sand or gravel. Finally, chlorine or another agent kills any remaining bacteria before the supply moves onward.
Because of this clear routine, each morning brings fresh water without risk. Science working quietly beside machines makes daily safety possible. Life feels easier when basics like this just work.
Flowers make more flowers - that much might surprise some. People aren’t the only ones who have offspring; green things do too. With blooming plants, most of the work takes place using what scientists label sexual reproduction.
Seeds begin to form when certain areas inside a bloom take part in the task. A straightforward look at how flowering plants reproduce sexually makes it clearer. What happens starts deep within the structure where life begins quietly. Step by step, pieces interact without noise or flash. Inside each blossom, tiny elements meet in silence. Growth follows after quiet meetings between pollen and ovule. These moments matter more than they seem. Hidden parts do their work without show.
Pollen lands on a sticky surface where life begins anew. Inside each bloom, tiny chambers hold dust that travels by chance. One part reaches out while another waits below. Together they make seeds without needing help from elsewhere.
Inside the bloom, the stamen handles pollen production. One piece lifts the tip high; another forms the base support
Pollen grains contain the male reproductive cells.
Inside the flower, the pistil handles seed making. Made of three pieces working together - sticky top catches pollen, middle tube moves it down, base swells into a pod where seeds grow
Female reproductive cells are found inside the ovules.
Pollen moves when it jumps from one part to another. That shift happens between spots made for the job. Movement kicks off once dust-like stuff drifts where it needs to go. The trip begins at a point that holds it, ends where it sticks. Travel starts small, finishes with connection.
Pollination can happen through:
Once pollen lands on the stigma, fertilization starts. Not until that moment does the plant proceed. Only then do cells react. From there, growth unfolds. Without this step, nothing follows. After contact is made, changes happen inside. Soon, tiny structures grow downward. These lead toward ovules deep within. The journey begins with a single grain.
Falling past the sticky tip, the grain sends out a thread that pushes through the neck of the flower toward its base. From there, the sperm moves along this path until it meets the egg within the seed chamber.
Fertilization gets its name from this merging.
After fertilization:
A tiny living part hides inside each seed, known as the embryo. Given the right environment, out pokes a sprout, turning slowly into a fresh green being.

Pollen comes from the stamen, while the pistil catches it during pollination. Once joined, cells meet inside the flower instead of staying apart. Seeds begin to grow after that mix happens deep within the plant. Fruits follow soon afterward, shaped by what took place earlier.
A single bloom holds more than color and scent. When you spot one, think again - its role goes beyond looking pretty, quietly working to help plants survive through generations.
One moment you flip the switch - suddenly, light fills the room. That quick glow comes from something small but clever: an electric loop doing its job. This loop creates a route so energy can move, turning on things we use every day. Bulbs brighten, fans spin, chargers work - all thanks to that steady flow guided by a clear path.
A single loop of wire can carry power when connected properly. We look at how that setup moves energy without confusion. Starting small helps see what happens step by step. This basic design shows flow using only essential parts.
A loop made of basic parts lets electricity move out from a battery, travel via an appliance, then return where it started.
Only when the route closes does current move through. A gap anywhere along it brings everything to a halt.
A simple circuit usually has four main parts:
Close the switch, then the loop finishes. From the battery, electrons move along the metal paths. Current reaches the bulb - light appears. A spark lives when power arrives.
Open the switch, power cuts - light dies. Break happens, flow halts because contact loses. Bulb shuts when electrons quit moving since path ends there.
Flow of electricity demands a loop without breaks. That setup goes by the name closed circuit. When gaps appear in the route, the term shifts to open circuit.
Simple circuits are found in:
Few realize how those hidden wires in walls follow a pattern - simple at heart, yet built up into something busier. A quiet echo of the original concept lives inside every circuit.
A basic loop of electricity forms every electronic setup you know. Power begins it, cables carry it, something uses it - sometimes a switch steps in. Close that path, current moves, gadgets wake up.
A single loop of wire shows how power moves. From here, everything else builds - no gadget too complex to trace back to this moment.
Lights glow, phones charge, machines hum - electricity makes it happen. Yet names like current and voltage pop up a lot in conversations. Puzzled faces appear when learners try sorting them out. Same thing? Not quite. One pushes, the other flows. Confusion slips in easily. What one measures, the other behaves. Clear lines blur without care. Each plays a role, yet neither replaces the other. Questions linger where answers should sit.
Here’s how current and voltage differ, explained plainly. What one moves, the other pushes. Flow comes from charge in motion, while pressure drives that flow. One measures movement of particles, the other measures push behind them. Think of it like water: amount passing through versus force behind it. Not the same thing, even if linked closely.
Flowing electrons move through a conductor when electricity travels along a metal path. Charges pass step by step inside thin strands meant to carry power quietly forward.
Faster flow inside the tube means more movement. That push behind the liquid? It’s what we call current.
Flow of electricity through a wire shows up as current. That movement gets counted in amperes. One ampere means a specific amount of charge passes by each second. The higher the number, the more charge travels. Measuring it helps understand what the circuit is doing. Tools made for this job give clear readings. Without such tools, guessing would be necessary. Yet precise values matter when things connect together. So checking current stays common practice.
More electrons moving along the wire means a stronger current. A weaker current happens when there are fewer electrons passing through.
A push of electricity moves electrons along a wire because of voltage. It acts like pressure guiding tiny particles where they need to go. Without this force, electron movement stops entirely. The stronger the shove, the faster those bits travel. Energy flows only when such tension exists between points.
Picture water moving through a hose - voltage acts much like the push behind it, guiding flow along the path.
A push on electric charges - how strong it feels - that's what voltage shows. Measured using volts (V), it gives a clear picture of force at work. From battery terminals to wall sockets, this measure appears everywhere power moves.
It takes voltage to get electrons moving; without it, current stops dead. A push is needed for flow - no force means nothing travels.
In simple words:
Pushing comes from voltage, flow happens with current.
A steady flow requires each of them to play its part. Without one, the whole thing stops moving forward.
What happens to current when voltage changes? That idea comes from Ohm’s Law. It shows how the two are linked. Voltage shifts push current one way or another. This pattern stays steady under certain conditions. The rule puts numbers to that behavior
current equals voltage divided by resistance
If voltage goes up while resistance remains unchanged, the flow of current rises too. What happens is a higher push leads to more movement through the circuit. When electric pressure climbs, so does the amount moving past any point each second. More volts with steady opposition result in greater current strength.
Electricity flows because of current and voltage - both matter a lot. Charges move; that movement is what we call current. Pushing them along? That job belongs to voltage. Without this push, nothing runs in a circuit.
When you think about plugging in a charger, picture voltage as the push behind tiny particles moving. Every time a light turns on, those particles are already traveling through metal paths inside walls. What makes gadgets run lies in that invisible motion driven by force and flow. Instead of just seeing switches flip, notice how energy finds its way quietly. Powering things happens because one part pushes while another carries along. Behind every working tool sits this quiet teamwork happening out of sight.
Water deep in the ground finds its way upward - how does that happen in trees hundreds of feet high? Leaves cook up nutrients using sunlight, yet those goods show up in roots without any visible delivery path. Stationary they seem, these plants, still rivers flow within their veins nonstop. Movement never stops beneath bark and stem.
Moving stuff like water and food inside plants uses tiny pathways built just for that job. Picture how it works without making it too hard. These channels pass what the plant needs from one spot to another, kind of like roads branching out under bark and leaves. Each part gets its share because the system spreads things where they belong. Think of roots pulling in moisture, then sending it up through quiet pipes hidden in stems. Leaves ship energy made by sunlight down to roots using separate lanes. Nothing moves randomly; each route has a purpose. Even flowers rely on these deliveries when growing. The whole thing runs without help once everything is in place.
From roots up, one tissue moves water through the plant. Another carries food made in leaves down to other parts. These systems work separately yet stay connected throughout growth
Inside the plant, these tissues serve as channels for transport.
Water moves up through the xylem, bringing along minerals from soil. From roots onward, it travels toward leaves because pathways inside allow steady flow. This transport system works quietly behind the scenes. Each part gets what it needs just in time.
This is the way it functions:
Water escapes through small leaf openings known as stomata, a process named transpiration. From there, an upward tug forms, moving moisture high into the air, sometimes hundreds of feet up, inside massive trees.
Fine tubes inside plants carry stuff up - roots push it toward the green tops.
Fresh under sunlight, leaves cook up meals with help from air and moisture. Yet every bit of the plant - roots, stems, the whole structure - depends on that supply.
From the leaves, sugars travel through phloem to reach different areas of the plant. Moving these nutrients is what translocation means.
Sometimes up, sometimes down, phloem moves food where the plant requires it. Xylem does not do this.
Without transportation:
Without it, the plant would struggle to survive. Movement inside delivers what's needed. Through tiny channels, substances reach every part. Life continues because flow never stops.
From deep down, roots send up water through xylem vessels. Moving sideways then upward, dissolved minerals travel along with it. Food crafted in leaf cells takes a different route entirely. Phloem tubes carry that energy-rich sap outward. One system rises, the other spreads. Together they keep every part fed.
Deep within, while leaves stay motionless, fluids move without pause through hidden channels. Nature built this quiet network - clever, steady, doing its job without show.
Each breath slips in without a thought, doesn’t it? Your lungs move on their own, moment after moment. What makes this quiet rhythm tick - have you pictured that before?
Oxygen moves into the body because of how the breathing parts work, while carbon dioxide gets pushed out at the same time. Survival drops fast if this process stops - just minutes matter when cells need air. A closer look makes it clearer, even if science feels tricky at first.
Our respiratory system includes:
From the nose down, every piece fits into how we take air in. Breathing relies on each section doing its job, quietly. Without one link, the chain stumbles, slowly. Air moves because pieces line up, just so. How it works depends on all parts showing up, always.
Into the nose flows air when breathing begins. Tiny hairs work there, catching dust while mucus holds back germs.
From there, air moves along the windpipe before branching into the lungs via structures known as bronchi. Tiny sacs named alveoli form at the end of ever-smaller pathways split off from those main tubes inside each lung.
Downward slides the diaphragm - a muscle beneath the lungs - when you breathe in. Chest cavity widens because of it, giving room for the lungs to stretch out.
Wrapped around each alveolus is a net of microscopic veins known as capillaries. From the air inside, oxygen moves across into the bloodstream here.
Breathing out carries carbon dioxide away when it shifts from blood to tiny air sacs. That shift? It’s what people name gas exchange.
From there, oxygen-filled blood travels everywhere inside you.
Breathing out pushes the diaphragm up, so the lungs squeeze tighter. From there, carbon dioxide escapes via mouth or nose.
Breathing in, then out, goes on without pause - it does not stop during sleep.
Inside each cell, food turns into energy when oxygen steps in. That change has a name - cellular respiration. If there is no oxygen around, the body starts to struggle.
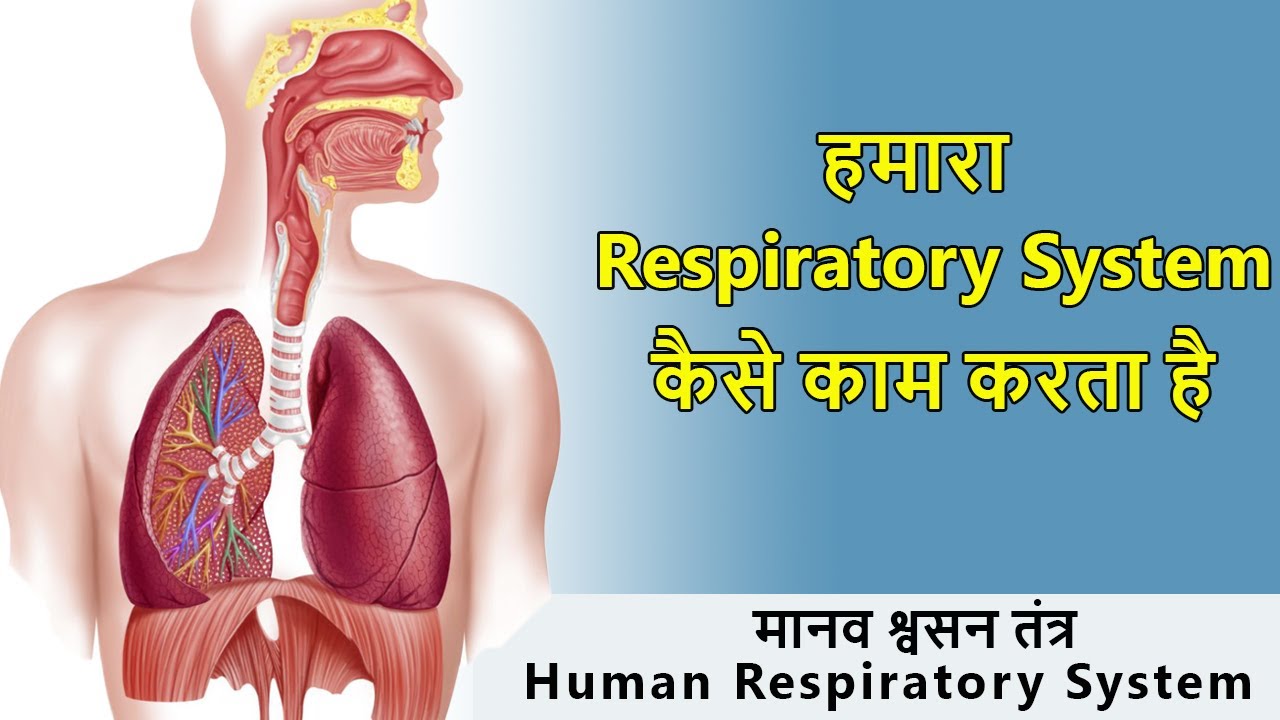
Breathing kicks off when air slips through the nostrils. After that, it travels down a network of tubes ending deep inside the chest. Oxygen sneaks into blood while old gases slip out through thin sacs. Every breath runs on quiet precision, unseen but never stopping. Parts link together without fuss - each one doing its share.
When air fills your lungs again, think about it - that quiet rush of oxygen through winding tubes is pure magic. Each breath slips past tiny gatekeepers, moving silently into hidden corners where life hums along unseen.
Every day brings shifts we notice without thinking twice. Melted ice disappears puddle by puddle. Burnt paper curls at the edges, blackening fast. Milk sours slowly, thickening into something new. Water jumps to steam when heated long enough. Yet here’s a thought - do each of these transformations follow the same path? Scientists sort such differences into two broad lanes: one where matter reshapes but stays true, another where it becomes entirely different inside.
Here’s how they differ, explained simply yet differently each time. What sets them apart becomes clear when you look closer. One thing leads to another without confusion. Each point connects but stands alone. This way of seeing helps without overwhelming. Seeing it this way makes the contrast pop. Not every detail needs emphasis - some just fit.
A shift happens when something alters how it looks - maybe bending, breaking, or melting - but what it's made of stays unchanged. Put another way, the stuff inside doesn’t turn into anything different.
Examples of physical changes:
Every time, it's the same stuff underneath. Think ice, water, then steam - each one just water wearing a different face. The form shifts: sometimes hard, sometimes fluid, sometimes invisible. State moves between frozen, flowing, or floating.
Frozen water turning to liquid might later become solid once more. Most shifts in form can go backwards.
Burning wood creates ash, a totally different material. Once changed like this, it stays that way most times. The result has traits the original never had. Reversing such shifts proves difficult nearly every time.
Examples of chemical changes:
Burning paper leaves behind ash along with smoke rising up. Once changed, the ash stays that way forever after. This one-way shift marks it clearly as a chemical process instead.
Signs of a chemical change may include:
Matter might shift shape without becoming something else - those shifts tend to bounce back. When stuff turns into a different material, it rarely flips to how it was before.

Fires crackle, ice melts - each moment holds shifts we barely notice. Shape might shift without altering what something truly is. Yet when molecules restructure, out pops a fresh kind of matter altogether.
What sets them apart makes things clearer - from how plants grow to why soap cuts grease. Not locked in labs, real science lives in raindrops, rust, morning coffee. It moves through moments most overlook.
What happens when your mouth puckers after biting into a citrus fruit? A slick film on skin after washing hands might give another clue. Behind moments like these lies something quiet but constant - how certain materials behave in water. Think of kitchen staples, bathroom bottles, remedies in cabinets. Each plays a role shaped by chemistry hiding just beneath touch or taste.
Picture this - clear, step by step. Not hard, just straightforward. A bit at a time, things start making sense. With each part, it clicks a little more. One idea follows another, smooth like that. Nothing fancy, only what matters. Slowly, the whole picture comes into view.
Bitter lemons hint at what acids often feel like on the tongue. When they meet metal, changes begin slowly. Water wakes them up, setting charged bits free. Those tiny H⁺ pieces drift where wetness flows.
Common examples of acids include:
Fizzing red when touched by blue paper - that's how researchers spot acids. Careless handling risks harm because powerful ones eat through materials fast.
Bitterness marks them, a soapy glide on skin. Slip into water, they push out hydroxide ions (OH⁻). Their presence felt through sharp flavors and slick touch.
Examples of bases include:
Red litmus paper shifts to blue when touching bases. Just as with strong acids, powerful bases may burn skin, so care is needed during use.
A splash of vinegar meeting baking soda shows what happens when acids meet bases - neutralization kicks in. Out comes salt, along with water, quietly forming from the mix.
For example:
Hydrochloric Acid Reacts with Sodium Hydroxide to Form Salt and Water
Not every salt tastes like what we sprinkle on food. From acids meeting bases, chemical salts emerge - different entirely.
Away from neutral, things tilt toward sour or soapy - that’s what scientists track using numbers between zero and fourteen called pH.
A single drop of water sits right at 7 on the scale - neither pushing nor pulling in either direction. Its balance gives it no tilt toward acid or base.

From vinegar to soap, chemicals shape routines without most people noticing. Sour tastes often mean an acid is present, one that pushes out hydrogen particles into liquids. Bitter hints? That usually points to a base doing its thing by releasing hydroxide bits. When these two meet in water, they team up quietly to build salts through quiet transformations.
Take a look at how acids, bases, and salts pop up everywhere - baking a cake, treating an illness, even cleaning dishes. Lemons sting cuts because they’re acidic; soap slips through fingers since it’s basic; table salt forms when those two clash quietly in water.
Feel that chill when you first grab a cold doorknob in winter? Same idea, just backwards - heat slips out of your hand into the metal. Touch drives this shift, no space needed between surfaces. Picture warmth sneaking through solids like walls, pans, or sidewalks under sunlight. Happens quietly, without wind or motion, just molecules nudging neighbors. You’ve seen it while stirring soup, where the handle slowly warms up too. Energy creeps along materials until balance shows up.
Let’s understand how conduction works in a simple and clear way.
When objects touch, warmth can move from the warmer one to the cooler one. This transfer takes place mostly in solid materials.
Heat moves between touching items, starting at the warmer one and heading toward the cooler. Only stops when temperatures match, guided by that initial difference. The transfer persists - driven entirely by imbalance - till warmth levels out across both.
Everything around you consists of small bits, such as atoms or molecules. Movement never stops in these little pieces.
When a solid object is heated:
This is how warmth spreads across stuff while bits stay put. Energy alone shifts position.
One end of a metal bar warms up if it sits near a flame. Over there, far from the fire, the opposite tip begins to feel warm too - not right away, but soon enough. This shift happens because warmth moves within the solid itself, slipping from particle to particle across tight spaces.
Few substances let warmth move right through without slowing it down. They go by the name of strong heat transmitters.
Examples:
For this reason, metal frequently becomes the material chosen for kitchen tools used in preparing food.
Heat moves slowly through certain stuff. Things like that? They’re known as insulators.
Examples:
Heat moves slow through wood, which explains the choice. Plastic works much like that, staying cool while metal parts warm up. Hands stay safe because these materials resist temperature flow.

Heat moves through touch - that is how conduction works. From warm spots to cool ones, it travels inside solid things. Metals pass heat fast; on the other hand, wood or plastic slow it down. This kind of energy shift stays close, never leaping across gaps.
Heat travels through materials in ways we notice every day. From warming a pan on the stove to building engines, movement of thermal energy plays a role. Though basic at first glance, this transfer shapes much of what happens in physical systems. It quietly powers explanations behind common experiences.
Most people believe time never changes. A single second seems identical no matter where you are, correct? Oddly enough, reality says otherwise. When motion increases or gravity shifts, time stretches or shrinks - verified by today’s physics. Scientists name this odd effect time dilation.
What happens when time slows down? That idea pops up thanks to Albert Einstein’s bold thinking in physics.
Besides standing still, racing fast through space alters how quickly moments unfold. When close to something heavy, like a planet or star, clocks tick slower than they do farther away. Not every observer measures seconds the same way. Motion changes everything - even the flow of time itself. Far from large bodies, time moves faster than near them. Speed reshapes experience, even if people don’t notice it daily.
Faster you move, time slows down - gravity tugs at it too.
One piece of that puzzle shows up in Einstein’s work on relativity.
There are two main types of time dilation:
Close to light speed, motion stretches time for the traveler versus someone standing still. A moving thing feels seconds drag when racing near that ultimate limit. For the swift mover, clocks tick slower than they do for those at rest. Speed warps how fast moments pass when nearing light's pace. Time crawls on board while outside observers see everything normal.
Astronauts moving close to light speed might come back to find everyone else aged more. Time slows down when you travel that fast compared to those staying behind. The faster the trip, the bigger the gap between clock ticks out there versus home ground. So while years pass on Earth, only days go by aboard such a craft. That difference shows up clearly after returning from such extreme journeys.
Floating close to something heavy - say a planet or a black hole - makes clocks tick slower. Gravity pulls harder, so seconds stretch longer.
When mass gets heavy enough, it warps the world around it. Closer to such giants, clocks tick at a lower pace.
What happens when clocks tick slower? Experiments show it's real. Proof sits in data from high-speed particles. Gravity changes time, fact confirmed by atomic clocks on planes. Evidence piles up from satellites too. This isn’t guesswork - measurements match predictions every run. Reality bends, just enough to notice.
Imagine clocks on satellites ticking just a bit off because they move fast while sitting high above Earth. Because of this, GPS and similar networks adjust their timing to stay precise. Without those tweaks, positions shown would drift more than expected.
Faults uncorrected, GPS accuracy slips fast.
Reality shifts when time stretches. Because space bends along with it, nothing stays rigid. How moments slow reveals a linked existence - where one thing tugs, the other follows.
Beyond Earth's edge, it shapes how we explore the cosmos, dig into star science, yet reveals secrets of intense things such as black holes.

Slowing of time happens when speed increases or gravity gets intense. Not fantasy - scientists observe it. Predicted by Einstein’s ideas about space and motion. Real results show clocks tick slower under these conditions.
Time stretching might sound odd - yet it shows how much stranger the cosmos turns out to be compared to old beliefs.
Out there, space keeps stretching - though not merely growing, it speeds up as years pass. Scientists did not expect that twist when they found it in the 1900s. Something must be driving galaxies away with rising force. That something remains a deep puzzle today: dark energy.
Something unseen pushes space outward faster every day. Though it stays hidden from tools and eyes, experts say nearly everything out there is made of this push.
Something unseen pushes galaxies apart, faster over time. This push comes from dark energy - invisible, silent, never glowing. Instead of clumping like stars or dust, it spreads evenly through empty stretches. Scientists notice its effect by how quickly distant points drift away. Though it shapes the cosmos, it refuses to touch normal stuff. Light passes right through without change. Instruments catch only the aftermath, never the thing itself.
Most of what's out there isn't stars or planets at all. Roughly two-thirds of the cosmos is something invisible called dark energy. Everything visible ends up being just a tiny piece of the whole.
Far off stellar explosions caught researchers by surprise near the end of the twentieth century. Not slowing as expected, space itself began stretching faster, contrary to earlier assumptions.
Scientists began suggesting dark energy was responsible for the speeding up, after this find came to light.
At first, the idea of space spreading out came from the Big Bang model - yet speed picking up shocked everyone.
Figuring out dark energy keeps scientists busy. Empty space might hold a kind of energy, some think. Gravity could behave in ways we just don’t get yet.
Filling space without gaps, dark energy gains influence when the cosmos stretches further.
Though people mix them up, dark energy isn’t the same as dark matter. One pushes space apart, while the other helps hold galaxies together through unseen mass. What seems similar turns out to act in opposite ways across the universe. Their names sound alike, yet their roles could hardly be more unlike
Most of what fills the universe comes from these two working together.
One big question shapes how we see space: what happens next? Could everything keep stretching without end? What if stars grow distant until light fades everywhere? Researchers look at signals from deep space to piece together clues.

A force we cannot see pulls at everything, quietly guiding what comes next. From empty space, something pushes outward - no light, just effect.
With every new image from deep-space observatories, answers about dark energy seem just out of reach. For now, its mystery remains - a quiet hint that much of the cosmos is still unknown.
Sometimes you tilt your head back under dark skies, thinking about space past our world. Earth itself? Just one tiny piece inside an immense setup known as the solar system. Hanging in place because of invisible pulls - this area holds stars like the Sun, large round planets, smaller orbiting moons, scattered rocks named asteroids, even icy travelers called comets. Everything stays linked without touching.
Picture Earth spinning fast while planets drift beyond the sun’s glow. A path unfolds past rocky worlds near bright heat. Then giants appear - huge spheres wrapped in swirling clouds. Their moons circle like quiet companions in dark space. Sunlight travels far before fading into cold stillness. Each object moves in rhythm without sound or rush. Ice hides where light barely reaches every few years. Tiny bodies gather where gravity pulls them slow.
Away from Earth, a group of space bodies circles a bright star called the Sun. This glowing core sits in the middle, sending warmth and brightness across vast distances to everything moving around it.
Around 4.6 billion years back, our solar system began inside a massive swirl of gas and dust. Yet it wasn’t sudden - slow pulls of gravity shaped what came next. Over time, clumps grew where space thickened. Then light sparked at the center. Elsewhere, bits collided, stuck, built more. So planets emerged piece by quiet piece.
A giant sphere of burning gas makes up the Sun. Because it fuses atoms deep inside, sunlight travels across space. Life here depends on that warmth reaching our planet.
Floating through space, each planet stays put because the Sun pulls them close. Instead of drifting off, they circle around steadily, held by that strong pull. Without it, everything would just fly away into darkness.
Eight big planets circle the Sun, sorted into two families
Firm ground covers these worlds, though they’re less wide than others. Their size runs shorter compared to giants floating out there.
Floating far out in space, these worlds pack huge sizes while built mainly from swirling gases and frozen stuff.
Besides the planets, the solar system also contains:
What ties everything in space? Gravity does. Planets stay in line because the Sun tugs on them hard. Without that pull, paths would go wild. Orbits remain steady thanks to this unseen grip.
A spin around an invisible line runs through every world in space. This turning creates light periods followed by dark ones. Night follows day because of how each globe moves. Rotation never stops, so time keeps shifting across their surfaces.
Out beyond our world, watching the solar system reveals secrets of planet birth. Sometimes a moon or rock gives clues about where life could hide in space. Weather on Earth? That connects too - seasons shift because of distant movements we see up there. Knowledge grows when researchers watch these patterns over time.
Out there, space probes plus observatories keep scanning the heavens, revealing fresh findings. While machines orbit and travel beyond Earth, they spot things never seen before. Now, distant planets show up more clearly through advanced lenses floating above the atmosphere. Even so, every mission adds pieces to a puzzle we barely understand. From faraway moons to hidden asteroids, clues appear where scientists least expect.

Around the Sun everything spins - eight planets, dozens of moons, pieces we barely notice. This collection of space stuff? It's where we belong.
Out there among the stars, learning about the planets shifts how we see ourselves. Space exploration tugs at curiosity once people grasp what orbits near Earth. Wonder grows when facts replace mystery around neighboring worlds.
Beautiful yet huge, the cosmos holds secrets no one has cracked. Even after hundreds of years learning its ways, plenty about space stays unclear. Out there, unseen energies mix with odd messages drifting through stars. Surprise sits at every edge of what we think we know.
Let’s explore some of the most fascinating unsolved mysteries of the universe.
Most of what fills space cannot be seen, researchers say. Hidden stuff like this goes by the name Dark Matter.
Light passes right through dark matter without being seen, absorbed, or bounced back. Because of how galaxies move, scientists know something unseen must be pulling. Out of everything in existence, nearly a third might be this hidden stuff.
Still, its makeup remains a mystery.
What hides beyond our sight? Dark Energy, far stranger than dark matter, shapes the cosmos. Speeding up cosmic growth - this unseen push defies old ideas. Not seen, yet its pull stretches space itself.
Far from expected, a strange stretch of space came to light near the century's end. Though it might fill almost seventy percent of everything, what dark energy actually is still slips through science's fingers.
Starting roughly 13.8 billion years back, the universe kicked off according to the Big Bang idea. Before that moment though - what was there?
Could a different universe have existed? Was time real at that point? Research continues through powerful telescopes and studies in subatomic particles.
Out there, tucked between stars and silence, sits a question people keep asking. Billions of galaxies swirl through space, each holding countless worlds. Life on our planet proves it can happen at least once. So why not again, elsewhere, under different suns? Trillions of chances stretch across the dark - maybe one holds something breathing.
Out there, researchers scan the skies for messages from distant worlds while tracking far-off planets circling other stars. Still, not one sign of another living thing has shown up - nothing solid at least.
Inside black holes, gravity pulls so hard nothing slips free - not even light. Their hidden cores? Still unknown.
Here sits the edge of a black hole - named the event horizon. Once crossed, what slips past appears lost forever. Peering into these giants could reveal how space, time, and pull truly behave.

Out there, beyond what we see, questions pull at every corner of space. Not just stars but hidden stuff - stuff that bends light without shining - a mystery shaping galaxies. Black holes? They swallow everything, even time seems to twist near them. Then again, somewhere far off, life might blink back at us from a distant world. Each clue comes slow, pieced together by minds refusing to look away.
What keeps science thrilling isn't knowing everything - it's chasing what lies beyond. Each answer pulls us into deeper curiosity, opening doors we didn’t see before. Wonder grows not in facts settled, but in mysteries stirred along the way.






 Ensure TV
Ensure TV








